Absorption is the process in which one or more components are removed from a mixture of gases, this is done to remove any impurities present within the mixture to achieve a separation in which aims to provide a pure specie of gas, (Richardson et al, 2002). Absorption reactions are typically housed within a column.
There are two main types of absorption processes: physical absorption and chemical absorption.
Physical Absorption
Physical absorption of a gas mixture will come into contact with a liquid solvent (known as the absorbent, which enters from the top of the column) wherein which some components of the gas will selectively dissolve into the liquid (known as the absorbate, which enters from the bottom of the column) via interfacial mass transfer (between boundary layers of the liquid and gas mixture).
Example: Dehydration of natural gas (absorbate) using diethylene glycol (absorbent).
Example 1:
What parameters would limit the rate of diffusion in physical absorption?
Chemical Absorption
Chemical absorption is similar to that of the physical absorption where a gas mixture will come in contact with a liquid solvent however, there is an additional chemical reaction between the absorbent and the absorbate.
Example: Removal of acid gases (such as carbon dioxide and hydrogen sulphide), which is a reversible reaction and can be seen in equations 1.11 and 1.12 below, respectively.
(1.11)
(1.12)
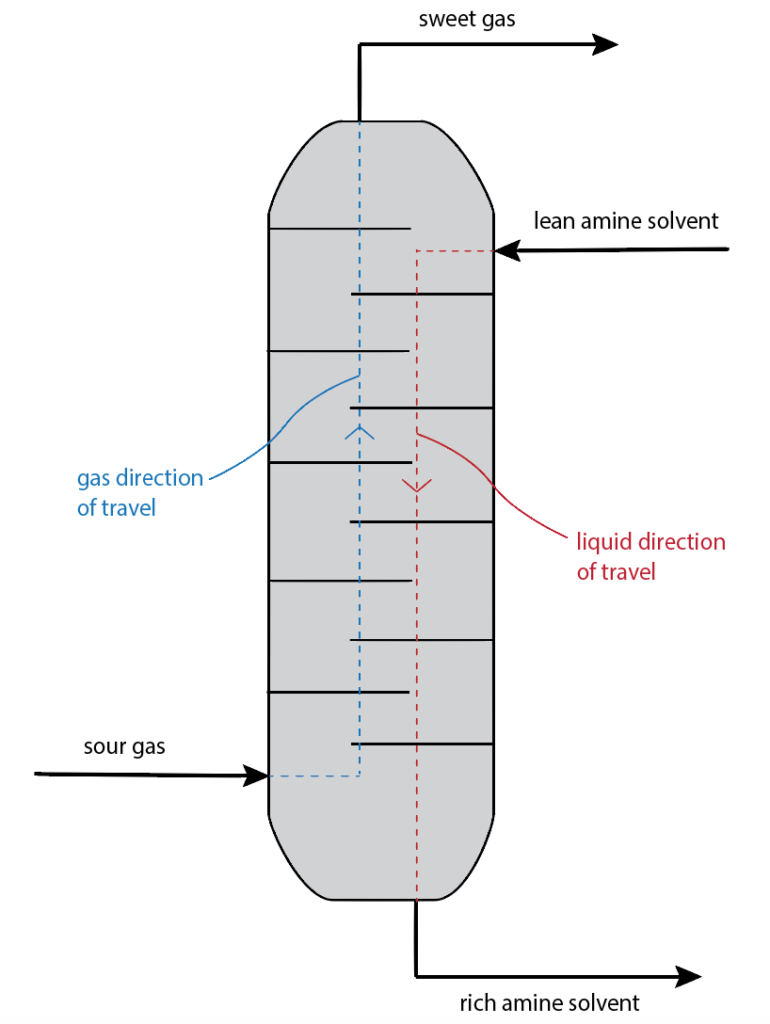
Figure 1: Shows a schematic drawing of an absorption column used to produce nitric acid, (“Wikipedia”, 2020).
Production of nitric acid and sulphuric acid, which is an irreversible reaction and can be see seen in equations 1.13 and 1.14 below, respectively.
(1.3)
(1.14)
Example 2:
What parameters would limit the rate of diffusion in chemical absorption?
Types of Columns
There are two main types of columns used in industry: packed and tray column.
Packed Column
In packed columns, the absorption columns are packed with material that enhances the contact between the absorbent and absorbate, where columns can be either packed with material randomly (such as Raschig rings) or in a specified structure, this can be seen in figure 2 below.

Figure 2: Shows an illustration of a packed column, where the upper half of the column has material structurally packed and the lower half of the column is randomly packed with material, (“Buffalo Brewing Blog”, 2020).
Between the two types of packing, random packing is much less expensive in comparison to that of structured packing whilst still being able to provide efficient contact between the liquid-gas boundary. However, structured packing is one of the better ways to distribute liquid across the entire cross-sectional area of the column as the liquid is spread so thin thanks to the structure packing. This then allows for a much smaller pressure drop throughout the column in comparison to that of the randomly packed columns, which in turn provides an increase in energy efficiency, (“MACH Engineering”, 2020).
Tray Column
Trayed columns consist of plates that force the liquids to flow back and forth horizontally as it travels down the bottom of the column, whilst the gas is passed through the holes of the tray, creating contact between the two phases.
Similar to packed columns, trayed columns are used to increase the contact area between the absorbent and absorbate, however, the major benefit of trayed columns is in its use for large diameter columns and would be able to handle large operational condition ranges.
Packed vs Tray Columns
| Packed | Tray |
| Has a lower liquid holdup | Better liquid distribution |
| Higher Recovery (better for batch distillation) | Much more column flexibility (e.g. additional feeds and side reboilers can be added) |
| Better for corrosive mixtures (packing material cheaper to replace than trays) | Higher residence time (much better for chemical absorption) |
| Better for smaller diameter columns | Better for low liquid rates |
| Lower Pressure drop (approximately 1/3 of that of tray columns) | Potential for higher turndown |
It can be seen from the table above that both types of columns have their advantages, however, when it comes to the design of columns, there are elements such as costs and sustainability of the unit operation that will help to decide which design is the best choice.
Reference
Buffalo Brewing Blog. (2020). Retrieved 1 August 2020, from https://www.buffalobrewingstl.com/pressure-drop/packedcolumn-flood-and-pressure-drop.html
Chuang, K.T. (2003). Tray Columns: Design. From https://www.semanticscholar.org/paper/Tray-Columns%3A-Design-Chuang/e207907eadf5209f2c74ded4f47c44bc35ebc85c
MACH Engineering. (2020). Retrieved 1 August 2020, from https://www.machengineering.com/random-packing-vs-structured-packing/#:~:text=Random%20packing%20uses%20a%20random,into%20a%20specific%2C%20fixed%20shape.
Richardson, J.F. Harker, J.H. Backhurst, J.R.. (2002). Coulson and Richardson’s Chemical EngineeringVolume 2 – Particle Technology and Separation Processes (5th Edition) – 12.1 Introduction. (pp. 656). Elsevier. Retrieved from https://app.knovel.com/hotlink/pdf/id:kt007FRVT3/coulson-richardsons chemical/absorption-introduction
Wikipedia. (2020). Retrieved 1 August 2020, from https://en.wikipedia.org/wiki/File:Simplified_absorption_column.png.

Adrian graduated with a Masters Degree (1st Class Honours) in Chemical Engineering from Chester University along with Harris. His master’s research aimed to develop a standardadised clean water oxygenation transfer procedure to test bubble diffusers that are currently used in the wastewater industry commercial market. He has also undergone placments in both US and China primarely focused within the R&D department and is an associate member of the Institute of Chemical Engineers (IChemE).

