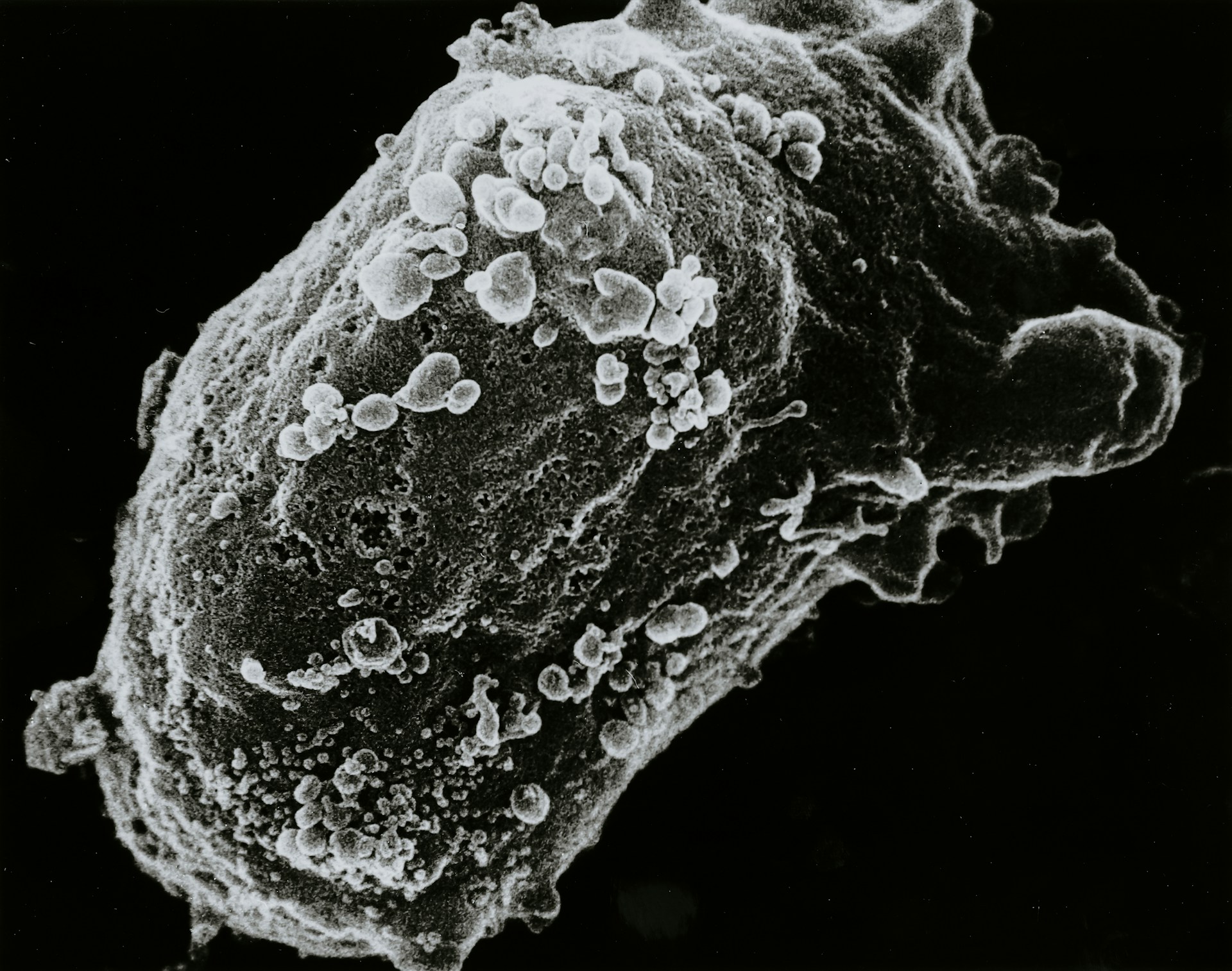
A research team at University of Málaga, led by Professor Antonia Gutiérrez and Dr. Juan Antonio García León, has identified prematurely aged astrocytes as a key contributor to the progression of Alzheimer’s disease. Their findings, published in the Journal of Neuroinflammation, suggest that cellular senescence in these support cells may actively drive neurodegeneration, particularly in individuals carrying the APOE4 genetic risk variant.
Caceres-Palomo, L., Sanchez-Mejias, E., Trujillo-Estrada, L., Perez-Moreno, J. J., Lopez-Oliva, E., Lim, T. E., DeFlitch, L., Chang, S. H., Kampman, L., Corces, M. R., Blurton-Jones, M., Moreno-Gonzalez, I., Pascual, A., Vitorica, J., Garcia-Leon, J. A., & Gutierrez, A. (2025). Human iPSC-derived APOE4/4 Alzheimer´s disease astrocytes exhibit a senescent and pro-inflammatory state that compromises neuronal support. Journal of Neuroinflammation, 23(1), 9. https://doi.org/10.1186/s12974-025-03607-z
Alzheimer’s disease has long been associated with amyloid plaques, tau tangles, and neuronal loss. However, increasing attention has turned toward the role of glial cells, which provide structural and metabolic support to neurons. Astrocytes, the most abundant glial cells in the brain, are essential for maintaining synaptic stability, regulating neurotransmitter levels, and supporting neuronal survival. The Málaga team’s work places these cells at the center of disease progression rather than at the periphery.
Professor Antonia Gutiérrez from University of Málaga stated,
“We have confirmed that these damaged astrocytes not only lose their ability to protect neurons but also adopt a pro-inflammatory profile that severely compromises neuronal survival.”
The researchers report that astrocytes in Alzheimer’s patients, especially those with the APOE4 genotype, exhibit signs of cellular senescence. Senescent cells remain metabolically active but lose their normal function and adopt a pro inflammatory profile. In this state, astrocytes accumulate DNA damage, show mitochondrial dysfunction, and release signaling molecules that can amplify inflammation within the brain. Instead of protecting neurons, they appear to compromise neuronal survival.
To investigate the mechanism in detail, the team generated astrocytes from induced pluripotent stem cells derived from patient skin samples. This approach allowed them to study human astrocytes carrying the APOE4 variant in a controlled laboratory environment. Compared with astrocytes derived from individuals without the high risk genotype, the APOE4 cells showed accelerated aging markers and reduced capacity to support neuronal health.
Importantly, the laboratory findings were supported by analyses of post mortem brain tissue from individuals with Alzheimer’s disease. In the cerebral cortex, approximately 80 percent of cells displaying premature aging characteristics were identified as astrocytes. This proportion was significantly higher than that observed in age matched individuals without the disease, reinforcing the conclusion that astrocyte senescence is not merely a byproduct of neurodegeneration but may contribute directly to it.
The APOE4 gene is widely recognized as the strongest genetic risk factor for late onset Alzheimer’s disease. While previous research has linked APOE4 to altered lipid metabolism and amyloid processing, this study connects the variant to astrocyte aging and inflammatory signaling. The findings align with a broader shift in Alzheimer’s research that views neuroinflammation as a central component of disease progression rather than a secondary consequence.
From a therapeutic standpoint, the results open a potential avenue involving senolytic strategies. Senolytic drugs are designed to selectively eliminate senescent cells or modulate their inflammatory activity. Although still under investigation in various age related conditions, such approaches could, in principle, be adapted to target dysfunctional astrocytes in Alzheimer’s disease. By reducing the burden of senescent support cells, it may be possible to preserve neuronal function and slow cognitive decline.
The study was conducted by the NeuroAD group within the Department of Cell Biology, Genetics, and Physiology at the University of Málaga, in collaboration with affiliated research platforms including IBIMA BIONAND and CIBERNED. The use of patient derived stem cell models, combined with validation in human brain tissue, strengthens the translational relevance of the findings.
While further research is required before clinical application, the identification of senescent astrocytes as an active driver of pathology marks a notable development in the field. Rather than focusing exclusively on neurons or protein aggregates, the work suggests that addressing the aging state of support cells may form part of a broader strategy to modify disease progression.
In a field where effective disease modifying treatments remain limited, redefining the cellular targets of therapy represents a meaningful shift. By placing astrocyte biology at the center of investigation, the Málaga team has contributed to an evolving understanding of how cellular aging intersects with neurodegeneration.

Adrian graduated with a Masters Degree (1st Class Honours) in Chemical Engineering from Chester University along with Harris. His master’s research aimed to develop a standardadised clean water oxygenation transfer procedure to test bubble diffusers that are currently used in the wastewater industry commercial market. He has also undergone placments in both US and China primarely focused within the R&D department and is an associate member of the Institute of Chemical Engineers (IChemE).