What Is Temperature?
Temperature is an objective measure of hot or cold and is measured using a thermometer. Temperature is used to determine if 2 system would be in thermal equilibrium in the event, they came into contact with each other through a diathermic wall and establish the energy flow.
There are two types of boundary:
- Diathermic: Thermally conduction, heat transfer occurs passing through freely.
- Adiabatic: Thermally insulting, no heat transfer to the surroundings.
An example of diathermic is when a hot object in thermal contact with a cold(er) object, resulting in heat flowing through the hot object to the cold object until thermal equilibrium is achieved where the heat flow stops and both objects are at the same temperature. For an adiabatic system, the heat in the hot object will not transfer to the cold object, as the heat in the hot object won’t go leave to it surroundings which would be the cold object (figure 1).
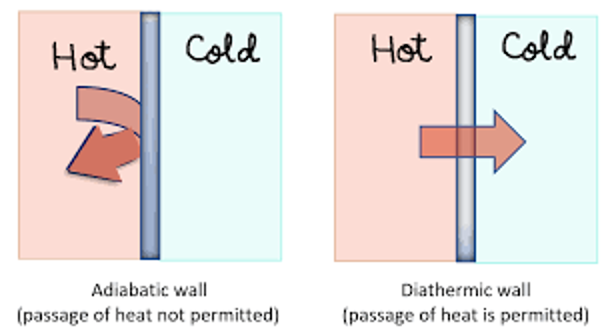
Figure 1: Adiabatic and diathermic wall heat transfer ( CIET, 2012).
What Is Pressure?
The pressure is afore exerted by a fluid per unit area, and the standard pressure is 1 bar which is measured with equipment that includes; barometers, manometers, fibre optic and piezoelectric. Most pressuring-measuring devices are calibrated to read zero when in the atmosphere, this is called gauge pressure.
In most equations absolute pressure is used, where p = 0 corresponds to a perfect vacuum and in a lot of equation this is called pabs. Also, pressures below atmospheric pressures are called vacuum pressures.
There is a relationship that relates these types of pressures (figure 2):
pabs = patm + pg & pabs = patm – pvac
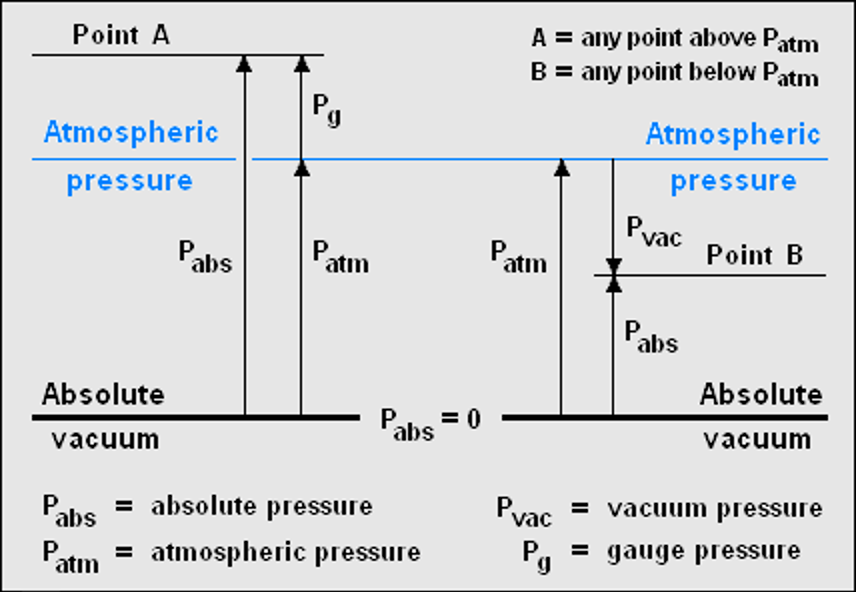
Figure 2: Relationship between gauge, absolute and vacuum pressure (Insta Tools, 2020).
Equilibrium
A system is in equilibrium with its surrounds when it does not tend to undergo spontaneous change. Whilst a system is in equilibrium, it will experience no changes when it is isolated from its surrounding.
Types of equilibrium:
- Mechanical equilibrium: No change in pressure within the system and doesn’t change with time.
- Thermal equilibrium: Temperature is constant throughout the system.
- Chemical equilibrium: Chemical composition is the same in the system and doesn’t change with time.
- Phase equilibrium: A system involving two phases and the mass of each system reaches an equilibrium level and stays within the system.
Describing A System
When describing a system, the state variables (p, n, T, V…) are independent of the system’s history and the number of properties required to fix the state of the system is given by the state postulate:
“The state of a simple compressible system is specified completely by two independent intrusive properties.”
Simple compressible system: If the system involves no electrical, magnetic, gravitational, motion and surface tension acting on it.
For a one-component system, all that is required is n (moles) and 2 variables, all other properties then follow:
V = f(n, p, t)
Example for notation:
A cylinder containing 2 moles of nitrogen gas at 1 bar pressure and a temperature of 25 oC (figure 3).
Notation: 2 N2 (g, 1 bar, 25 oC)

Figure 3: Cylinder containing nitrogen gas for notation example.
References
CIET. (2012). Physics-02. Retrieved from CIET: https://ciet.nic.in/moocspdf/Physics02/Unit08/keph_201201_eContent2019.pdf
Insta Tools. (2020). Difference Between Absolute and Gauge Pressure. Retrieved from Insta Tools: https://instrumentationtools.com/difference-between-absolute-and-gauge-pressure/

Dr. Adam Zaidi, PhD, is a researcher at The University of Manchester (UK). His doctoral research focuses on reducing carbon dioxide emissions in hydrogen production processes. Adam’s expertise includes process scale-up and material development.’



