What is Heat Transfer?
Energy can exist in numerous forms;
- Thermal,
- Mechanical,
- Kinetic,
- Heat,
- Potential,
- Electrical,
- Magnetic,
- Chemical,
- Nuclear.
The sum of these energy is the total energy and the sum of all microscopic forms is called the internal energy of a system.
Heat is a form of energy (sometimes called heat energy), that can be transferred between systems when there is a temperature difference. Heat transfer occurs from a higher temperature region to a lower temperature region, with heat transfer stopping when there is no longer a temperature difference.
There are three types of basic model that describe heat transfer, these are;
- Conduction
- Convection
- Thermal Radiation
Conduction
Conduction involves the transfer of heat from more energetic particles of a substance to less energetic particles, due to the interaction between the two particles (Figure 1). In solids conduction occurs due to vibrations of molecules and the energy transport by free electrons. For gases and liquids conduction occurs due to collisions and diffusion of molecules during their random motion.
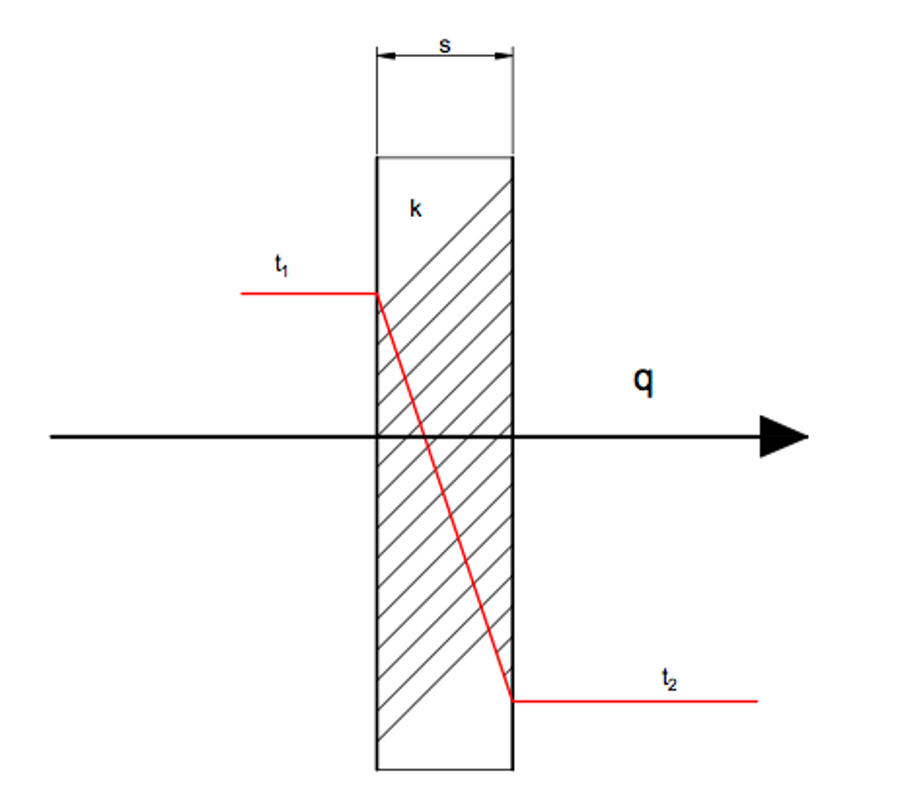
The rate of heat conduction through a plane is proportional to the temperature difference across the layer and the heat transfer area, and is inversely proportional to the layer thickness:
Rate of heat conduction ∝ (area x temperature difference) / thickness
Qcond = kA x (ΔT/Δs)
k – thermal conductivity a measure of a materials ability to conduct heat
A – Area of the material
The heat transfer area is the thermal conductivity multiped by the material area. A high thermal conductivity means that the material is a good heat conductor, and low thermal conductivity shows that the material is a bad conductor or an insulator. Heat is conducted from the higher temperature regions to lower temperature regions, with the temperature gradient becomes negative when temperature decreases with increasing thickness.
Convection
Convection involves heat transfer between a solids surface and a moving liquid or gas, and involves both conduction and fluid motion, with the greater fluid motion the greater the convection heat transfer (figure 2). If there is no fluid motion, then heat transfer would be purely by conduction.
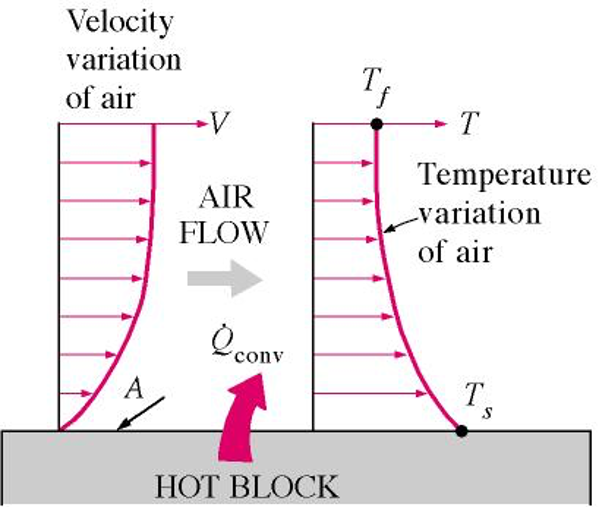
There are two types of convection, forced convection and natural convection (figure 3).
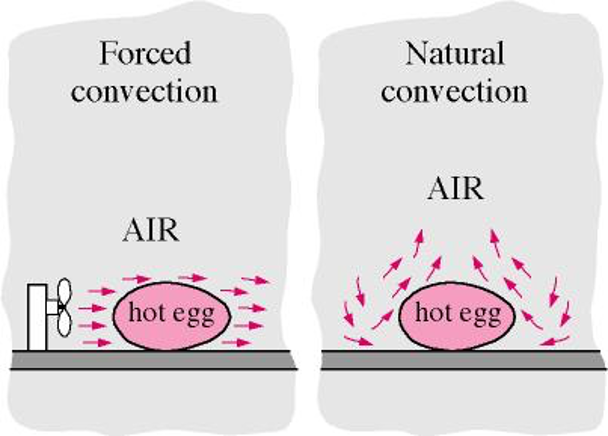
- Forced convection – forcing a fluid to flow over a solid surface,
- Natural convection – fluid motion is due to the buoyancy force that are induced by density differences due to temperatures variations in the liquid or gas.
Furthermore, a change in phase of a fluid is classified as convection as the fluids are in motion such as vapour bubbles during boiling or liquid droplets during condensation.
The equations for convection involved the surface area and convection heat transfer coefficient:
qconv = hA(Ts-Tf)
h – convection heat transfer coefficient (W/(m2-K))
A – surface area convection is taking place (m2)
Ts – surface temperature (K)
Tf – temperature of fluid away from the surface (K).
Thermal Radiation
Heat transfer by thermal radiation is done by electrometric waves and doesn’t require any medium and can pass through a vacuum (figure 4). Thermal radiation is caused by the random movement of atoms and molecules, with the movement of the charged protons and electrons results in the emission of electromagnetic radiation.
The hotter an object the greater the thermal radiation, with the temperature of an object affecting the wavelength and frequency of the radiated waves. As temperatures increase the wavelengths of the emitted spectra decrease, resulting in a short wavelength high-frequency radiation.
The Stefan-Boltzmann law is used to calculate thermal radiation:
P = e · σ · A · (Tr4 – Tc4)
- P = net radiated power;
- A = radiating area;
- Tr = temperature of the radiator;
- Tc = temperature of surroundings;
- e = emissivity;
- σ = Stefan’s constant.
Emissivity is as object’s effectiveness in emitting energy as thermal radiation and is a ratio, at a given temperature, of the thermal radiation from a surface to the radiation from an ideal black surface as determined by the Stefan-Boltzmann law (machinedesign, 2015). Stefan’s constant is determined by constants of nature:
σ = (2 · π5 · k4)/(15 · c2 · h3) = 5.670373 × 10–8 W · m–2 · K–4
k = Boltzmann’s constant;
h = Planck’s constants;
c = speed of light in a vacuum.
Common materials have lower emissivity values than an ideal radiator which has a value of 1.
References
claesjohnson. (2011). claesjohnson. Retrieved from Radiative Heat Transfer: Theory: http://claesjohnson.blogspot.com/2011/10/radiative-heat-transfer-theory.html
Engineering ToolBox. (2020). Conductive Heat Transfer. Retrieved from Engineering ToolBox: https://www.engineeringtoolbox.com/conductive-heat-transfer-d_428.html
machinedesign. (2015). What’s the Difference Between Conduction, Convection, and Radiation? Retrieved from machinedesign: https://www.machinedesign.com/learning-resources/whats-the-difference-between/document/21834474/whats-the-difference-between-conduction-convection-and-radiation
McGraw-Hill Higher Education . (2020). Convection Heat Transfer. Retrieved from McGraw-Hill Higher Education : http://www.mhhe.com/engcs/mech/cengel/notes/ConvectionHeatTransfer.html

Dr. Adam Zaidi, PhD, is a researcher at The University of Manchester (UK). His doctoral research focuses on reducing carbon dioxide emissions in hydrogen production processes. Adam’s expertise includes process scale-up and material development.’



