Batch Process
A batch process (figure 1) is a process whereby during the operation, no materials enter or leave the system during a period, then some material is left in the system this is called material accumulation and then the vessel must be cleaned between each batch. In industry batch reactors are used, when there is a range of products that have different reactants and required different conditions, thus the same reactor can be used. Examples of Batch reactors are used in; beverage processing, dairy products, pharmaceutical formulations and soap manufacturing.
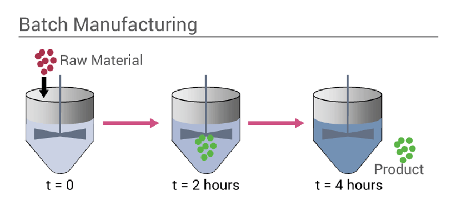
Figure 1: Batch process system (Cox, 2020)
Advantages And Disadvantages Of Batch Processes
|
Advantages |
Disadvantages |
|
Allows multiple products to be products from the same equipment, helping to reduce capital costs. |
Limited to a small scale. |
|
Flexible production rate due to different types of reactant and products can be used and produced |
Quality can’t be maintained and there will be variations from one batch to another. |
|
Easy to clean and maintain. |
The plant is idle for some time due to changes of batches and cleaning. |
|
Easy to scale up by simply adding more vessels. |
Labour intensive thus operating costs are higher. |
|
Quality can be maintained by checking of each batch. |
Usually only favourable for high-value products that are produced in small quantities such as perfumes |
|
Batch qualities can be easily traced which helps find any defective batches. |
Other Types of Batch Process
There are other types of batch processes such as, semi-batch (figure 2) which is a process that is neither batch nor continuous, and reactant or products in times are either added or removed. Examples of where semi-batch is used are in the pharmaceutical industry in tablet compression or roller compaction.
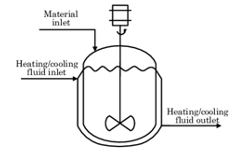
Figure 2: Semi-batch system (Zuluaga-Bedoya, 2014)
Another type of batch process is Fed-batch (figure 3), which during the operation materials enter intermittently or continuously into the process and the product remain in the reactor until the end of the run. Fed-batch is an operational technique used in biotechnological processes for the production of organic solvents and acids, as well as in fermentation.
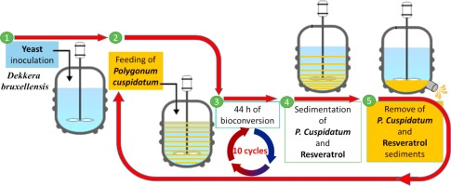
Figure 3: Fed-batch system example for wine yeast fermentation (Kuoa et al, 2017)
Continuous Process
For a continuous process (figure 4) as the process is ongoing materials are feed continuously into the system and products, by-products and waste stream the system without interruption. Unlike batch systems cleaning is infrequent and is done rarely when there is a plant shutdown or maintenance. Examples of continuous processes are; oil refining, chemical manufacturing, fertiliser production, power stations, wastewater treatment and many others.
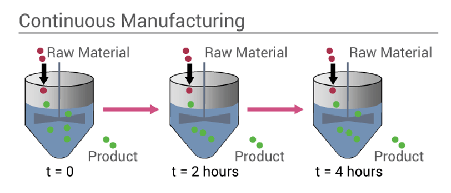
Figure 4: Continuous process system (Cox, 2020)
Advantages and Disadvantages of Continuous Processes
|
Advantages |
Disadvantages |
|
Cost advantages that companies can obtain due to their scale of operation (Economies of scale) can be released due to large scale applications being possible. |
Only allow for one type of product from the same equipment. |
| Easier to maintain quality as the product will be the same throughout as it is running continuously. |
Hard to clean and maintain as the process is running constantly. |
|
Easier to optimise reaction conditions against process efficiency
|
High capital costs due to lots of equipment being required. |
| Easier to recycle and recover heat |
Low flexibility in changing products, what the process is designed to produce is usually the only thing it can produce without any changes being needed, |
|
High process utilisation as the plant is constantly running |
Malfunctions anywhere in the process will mean the whole process must be shutdown |
| Not as labour-intensive compared to batch as the process works continuously so less human intervention required | |
| Control of the entire process is easier |
|
References
Cox, B. (2020). Will Continuous Manufacturing Mean Continuous Generic Delay? FDA Hears AAM’s Warning. Retrieved from Informa Pharma Intelligence: https://pink.pharmaintelligence.informa.com/PS122069/Will-Continuous-Manufacturing-Mean-Continuous-Generic-Delay-FDA-Hears-AAMs-Warning
Hsiao-Ping Kuoa, R. W.-S.-T.-C.-T. (2017). Pilot-scale repeated fed-batch fermentation processes of the wine yeast Dekkera bruxellensis for mass production of resveratrol from Polygonum cuspidatum. Bioresource Technology, 986-993.
Zuluaga-Bedoya, A. F.-M. (2014, November). Multi-stage control policies on batch suspension polymerization reactor. Retrieved from Research gate: https://www.researchgate.net/publication/276271068_Multi-stage_control_policies_on_batch_suspension_polymerization_reactor

Dr. Adam Zaidi, PhD, is a researcher at The University of Manchester (UK). His doctoral research focuses on reducing carbon dioxide emissions in hydrogen production processes. Adam’s expertise includes process scale-up and material development.’



